Patent Battles in the Age of Crispr
The CRISPR/Cas9 system (CRISPR) developed for gene editing only a few short years ago* has already revolutionized genomic engineering, and clinical trials for targeted cancer therapies have recently commenced in China and the United States.
CRISPR's power lies in its relative simplicity and precision, and it is expected to have profound implications for agriculture and the treatment of disease. No surprise, then, that the groundbreaking technology is already beset with controversy ranging from ethical issues associated with the technology† to a highly publicized and public US patent dispute.
CRISPR’s nucleic acid sequences are part of a naturally occurring bacterial defense system in which repeating sequences of genetic code are interrupted by “spacer” sequences. The spacer sequences represent remnants of genetic code from past viral invaders. Using enzymes of the CRISPR system, the bacteria snip out parts of the virus DNA and keep a portion of it behind to help them recognize and defend against the virus next time it attacks, much like an immune system.
This bacterial defense mechanism has been adapted by scientists to form a number of related gene-editing systems, yet the CRISPR/Cas9 system has been the main focus of attention to date. Here, two key molecules are used to modify a DNA molecule: guide RNA and the enzyme Cas9. Guide RNA (gRNA) is a piece of predesigned RNA sequence about 20 bases long, located within a longer RNA scaffold. The gRNA has RNA bases that are complementary to those of the target DNA sequence, which allow it to find and bind to that specific DNA sequence (Figure 1A). Cas9, which is in a complex with the gRNA, follows the gRNA to the same location in the DNA sequence. Together they bind to the target DNA site, a process that also requires the presence of a sequence called the protospacer adjacent motif that facilitates Cas9 binding.
Cas9 then makes a cut across both strands of the DNA, resulting in a double strand break (Figure 1B). At this stage, the cell recognizes that the DNA is damaged and tries to repair it. Scientists can then use DNA repair “machinery” to introduce specific changes in the host DNA sequence, such as a new mutation, a sequence addition, or a sequence deletion (Figure 1C).
CRISPR can be designed to target virtually any DNA sequence. So far it has been adapted to alter genomes that include yeast, worms, fruit flies, zebra fish, plants, mosquitoes, mice, monkeys, and human cells.
Patent Proceedings
Not surprisingly, CRISPR’s wide-reaching and powerful potential applications have led to significant interest in protecting the intellectual property associated with this technology. As with many important scientific advances, both individuals and research groups were involved in identifying the CRISPR system in nature, recognizing that it could have far-ranging applications, and then adapting it as a genome- editing technology. ‡
In June 2012 a group of researchers led by Jennifer A. Doudna of the University of California (UC), Berkeley, and Emmanuelle Charpentier of Umeå University in Sweden published a seminal paper in Science describing a CRISPR/Cas9 system that could cut DNA in vitro.5 By the time the paper was published, the researchers had already filed a patent application with the United States Patent and Trademark Office (USPTO) claiming methods and compositions for modifying DNA (the “UC application”).6
In December 2012, a second group of researchers, led by Feng Zhang of the Broad Institute, filed a patent application with the USPTO (the “Broad application”). Unlike the UC application, which described the use of CRISPR/Cas9 only in prokaryotic cells (unicellular organisms that lack a nucleus or organelles), the Broad application showed that the CRISPR/Cas9 system could be used in eukaryotic cells (those that have a membrane-bound nucleus and other organelles) to modify DNA in vivo.
While the Broad application was filed after the UC application, it requested and received accelerated examination and issued to patent first, in April 2014.7 The Broad patent claims cover methods of editing genes in eukaryotic cells using CRISPR/Cas9.
The UC application was filed under the “first to invent” patent system in the United States.
* For one a review of the timelines and scientists involved in developing CRISPR, see reference 1 at the end of this article. † The ethical issues associated with gene editing are not discussed here. For one review, See reference 2 for one perspective. ‡ Briefly, repeated sequences of 30 bases separated by spacers of approximately 36 bases were identified in an archaeal microbe by Francisco Mojica in 1993 (see reference3 ). Over a decade later, Mojica realized that CRISPR loci are part of an adaptive defense system that protects microbes against specific infections (see reference 4 ). Encouraged by these initial discoveries, scientists began to explore ways by which this natural process could be adapted to edit genes.
- 5Jinek, M., et al., “A Programmable Dual-RNA-Guided DNA Endonuclease in Adaptive Bacterial Immunity,” Science 337, Issue 6096 (2012): 816–821.
- 6Doudna, Jennifer, et al. US Patent Application No. 13/842,859. “Methods and Compositions for RNA-Directed Target DNA Modification and for RNA-Directed Modulation of Transcription.” 6 March 2014. https://patentimages.storage.googleapis.com/pdfs/ad6b5ca5819797f94952/US20140068797A1.pdf
- 7US Patent Publication No. US 8697359 B1. “CRISPR-Cas Systems and Methods for Altering Expression of Gene Products.” 15 April 2014. https://www.google.com/patents/US8697359
- 1Lander, Eric S., “The Heroes of CRISPR.” Cell 164, nos. 1–2 (16 January 2016): 18–28.
- 2Bosley, Katrine S., et al., “CRISPR Germline Engineering—The Community Speaks” Nature Biotechnology 33 (2015):478–486.
- 3Mojica, F. J. M. et al., “Transcription at Different Salinities of Haloferax mediterranei Sequences Adjacent to Partially Modified PstI Sites.” Molecular Microbiology 9, Issue 3 (August 1993): 613–621.
- 4Mojica, F. J. M., et al., “Intervening Sequences of Regularly Spaced Prokaryotic Repeats Derive from Foreign Genetic Elements,” Journal of Molecular Evolution 60, Issue 2 (February 2005): 174–182
While the United States has now switched to a “first to file” patent system, consistent with the majority of patent systems around the world, the older system awards a patent to the first person to invent a new technology. When questions arise under this system with respect to which party first invented a commonly claimed invention, administrative proceedings known as “interference proceedings” are held. These proceedings are now obsolete under the current system.
In early 2016, UC (and related parties) requested that a patent interference proceeding be initiated. They claimed that the Doudna/Charpentier team invented the CRIPSR/Cas9 system and that the disclosure in the Broad patent and related patents and applications that the system worked in eukaryotic cells was merely an obvious extension of Doudna and Charpentier’s work. Essentially, UC’s position was that both the UC application and the Broad patent were directed to the same invention and that the Doudna/Charpentier team were the first inventors. According to this rationale, the Broad family of patents and applications were invalid.
In a decision released on 15 February 2017, however, the USPTO’s Patent Trial and Appeal Board (PTAB) rejected the UC argument. The PTAB agreed with the Broad Institute’s position that one of ordinary skill in the art would not have reasonably expected that a CRISPR/Cas9 system as described in the UC application would work in eukaryotic cells. Thus, according to the PTAB, the UC and Broad applications are directed to two different inventions and there is no interference between the two patent families.
Because that issue was determinative of the interference, the PTAB declined to decide any other issues and terminated the interference. In summary, the PTAB found that:
- Broad provided sufficient evidence to show that its claims, which are all limited to CRISPR-Cas9 systems in a eukaryotic environment, are not drawn to the same invention as UC’s claims, which are all directed to CRISPR-Cas9 systems not restricted to any environment. Specifically, the evidence shows that the invention of such systems in eukaryotic cells would not have been obvious over the invention of CRISPR-Cas9 systems in any environment, including in prokaryotic cells or in vitro, because one of ordinary skill in the art would not have reasonably expected a CRISPR-Cas9 system to be successful in a eukaryotic environment. 10
Practically, this means that the Broad patent remains valid, and that the UC application can proceed.
At time of publication, the case has not yet been settled. On 13 April 2017, the UC filed an appeal of the PTAB decision to the United States Court of Appeals for the Federal Circuit, so there is a possibility that the decision itself could be overturned.§11 Even if the decision is upheld, it is still unknown what patent claims will ultimately issue from the UC family of applications.
§ The Broad Institute’s opening brief in the case is due 25 October 2017. ** Based on a USPTO patent application database search on 13 June 2017 for entries containing “CRISPR” in the title. The search results were not reviewed. It should be noted that patent applications are not typically published until 18 months after filing, so there may be many more applications pending that are yet to be published.
- 10United States Patent and Trademark Office. The Broad Institute, Inc., vs. The Regents of the University of California. 15 February 2017. https://drive.google.com/file/d/0B1Qqijz4aI6hdV9tdC1Zb05RcXc/view
- 11Noonan, Kevin E. “University of California/Berkeley Appeals Adverse CRISPR Decision by PTAB.” Patent Docs, 13 April 2017. http://www.patentdocs.org/2017/04/university-of-californiaberkeley-app
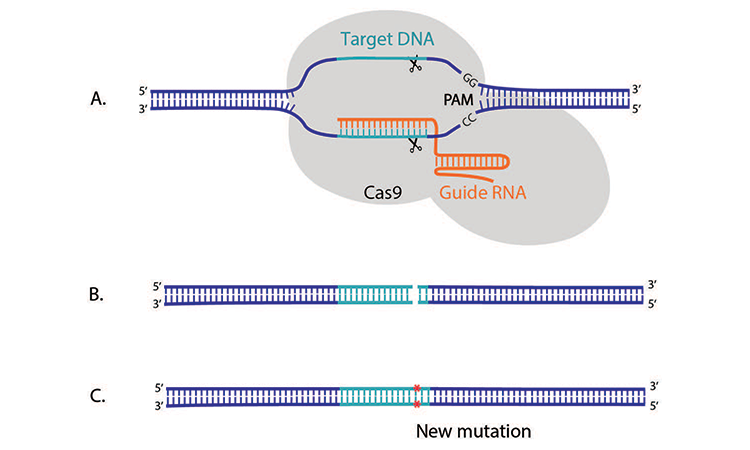
1B. Cas9 makes a cut across both DNA strands, resulting in a double strand break.
1C. This triggers cellular DNA repair, and enables the introduction of specific changes such as mutations in the target DNA sequence
High Stakes
If, as expected, CRISPR proves to have diverse applications in agriculture and medicine, the holder of any key patent stands to gain a pretty sum. In spite of the ongoing patent dispute, companies have moved quickly to license CRISPR patent portfolios, and several CRISPR biotechs went public in 2016 with initial public offerings worth $90 million and more.8 For third parties wishing to license the CRIPSRCas9 technology, the current licensing landscape remains murky at best.
Figure 1: The CRISPR/Cas9 system
For example, even if the Broad Institute’s patents covering the use of CRIPSR-Cas9 in eukaryotic cells (i.e., including human cells) are upheld, it is possible that the UC application will issue with broad claims to the use of CRISPR-Cas9 in any cell. Depending on the claims that issue for the UC patent, a third party hoping to edit eukaryotic cells using CRISPR (e.g., in the development of human therapeutics) would potentially need to license both the Broad and UC patents. In addition, both patent portfolios are already licensed to a number of different parties under a variety of arrangements including, in some cases, exclusive licenses, adding further layers of complexity.
Furthermore, it is likely that improvements and alternatives to CRIPSR-Cas9 will be developed. Searching the USPTO database for pending applications that include “CRISPR” in the title identifies 86 applications published in 2016 and 2017.** For example, alternatives that do not use the Cas9 enzyme may also work. In particular, the Cpf1 endonuclease has been shown to be a CRISPR effector. 9
Conclusion
The CRISPR system is an exciting, groundbreaking, and game-changing technology. It has already revolutionized laboratory science and, if expectations are realized, will have profound effects on the treatment of cancer and genetic diseases. In addition, the technology is expected to have big implications for food, pest control, and livestock, and will likely have many as of yet unforeseen applications. The patent battles for control of CRISPR technology are underway, but it will be some time before it is clear whether the victory will be shared or if it will be a case of “winner takes all.”
- 8Adams, Ben, “CRISPR Cuts Out $90M IPO bid as Another Gene Editing Biotech Goes Public,” FierceBiotech, 12 September 2016. http://www.fiercebiotech.com/biotech/crispr-cuts-out-90m-ipo-bid-as-another-gene-editingbiotech-goes-public
- 9Zetsche, Bernd, et al., “Cpf1 Is a Single RNA-Guided Endonuclease of a Class 2 CRISPR-Cas System,” Cell 163, No. 3 (22 October 2015): 759–771.


